Sterile Thermoforming Packaging Solutions for Pharmaceutical & Medical Devices

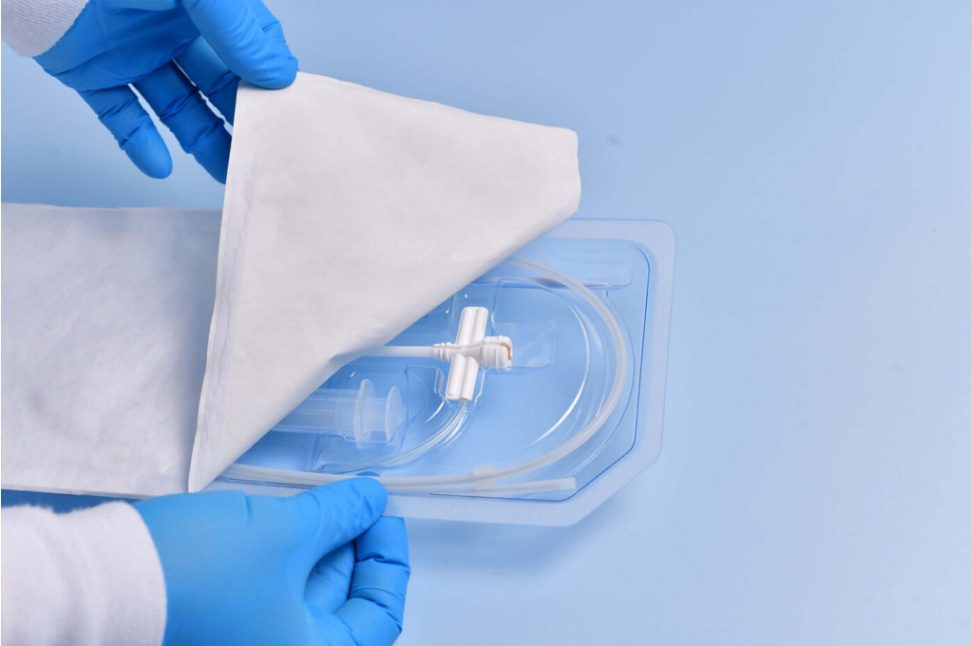
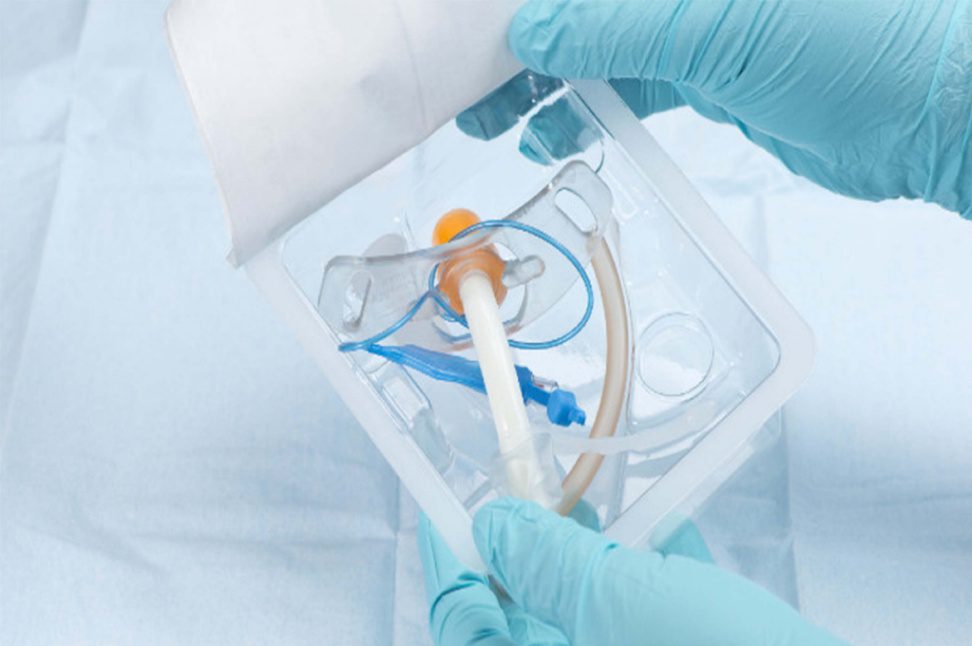
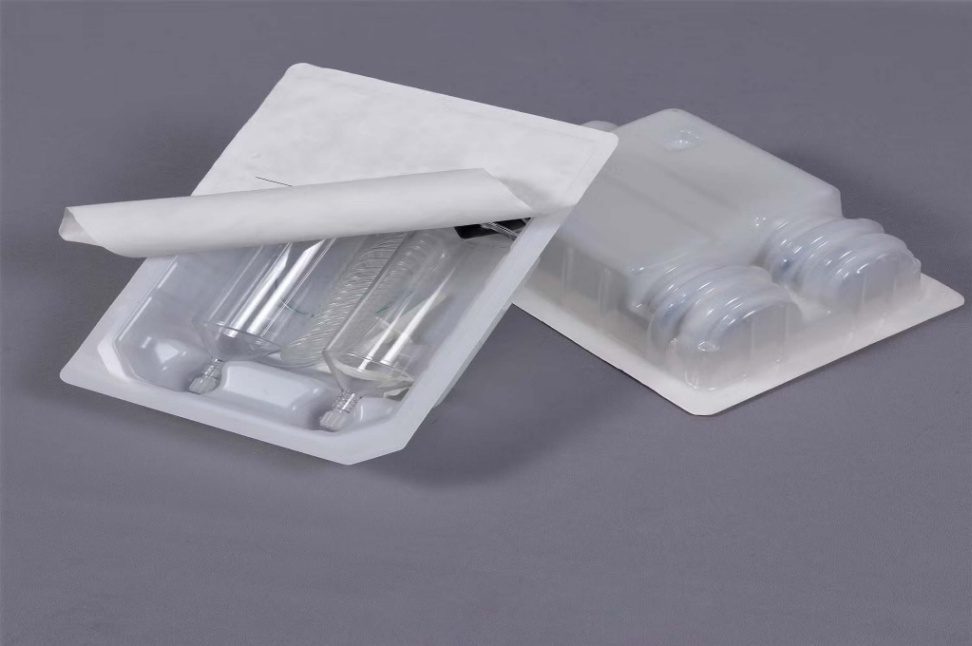
Advanced Sterile Packaging for Pharma, Biotech & Healthcare
Sterile packaging is essential for maintaining product safety, ensuring contamination-free environments, and supporting regulatory compliance. Trizen Packaging offers sterile thermoforming packaging solutions designed to protect pharmaceutical products, medical devices, diagnostic components, and surgical consumables.
Our sterile thermoforming trays are widely used in pharmaceutical manufacturing, medical device packaging, and aseptic processing environments.
These trays help manufacturers and healthcare providers maintain sterile integrity from production to patient use — ensuring reliable performance in all clinical environments.
Every sterile packaging solution is manufactured in an ISO 9001:2015–certified facility, ensuring consistent quality, process control, and compliance for medical applications.
Engineered Sterile Thermoforming Trays for Product Integrity
Sterile pharmaceutical and medical products require advanced packaging that prevents microbial ingress, physical damage, and environmental exposure. Trizen’s sterile trays are engineered with:
Sealable flange rims for sterile barrier sealing
Precision-formed cavities for accurate product fit
Anti-microbial compatible surfaces
High barrier protection against moisture and particulates
Tamper-evident design options
These features ensure sterility is maintained during filling, sealing, transportation, storage, and clinical use.
Material Selection for Sterile Barrier Systems
Trizen Packaging uses medical-grade materials suitable for sterile thermoforming packaging, including:
PETG: Ideal for sterile barrier systems and EtO/gamma sterilization
PP: Suitable for steam sterilization and chemical resistance
PET / APET: Strong and stable for surgical kits and device packaging
HDPE: Durable for protective and heavy-duty applications
These materials are compatible with EtO, gamma, e-beam, and steam sterilization processes, ensuring consistent sterile performance.
These materials ensure reliable performance of sterile thermoforming packaging solutions in regulated healthcare environments.
Applications Across Pharma, Biotech & Medical Care
Sterile thermoforming trays support a wide range of pharmaceutical and medical applications, including:
⦁ Injectable & Biologic Containers: Trays for syringes, vials, ampoules, and cartridges
⦁ Surgical Instrument Kits: Sterile, sealed trays for OR-ready instruments
⦁ Catheter & Tubing Sets: Protective packaging for vascular and respiratory devices
⦁ Diagnostics & Test Kits: Trays that maintain sterility for IVD components
⦁ Implantable Devices: Packaging for orthopedic, ophthalmic, and cardiology implants
⦁ Aseptic Filling Lines: Trays for automated sterile production
These trays improve operational efficiency while ensuring contamination-free handling across healthcare and manufacturing environments.
Custom Sterile Thermoforming Tray Design & Validation
CNC-engineered sterile mold design
Rapid prototyping for sterilization validation
Custom flange and sealing surfaces
Compatibility with Tyvek®, films, and laminates
High-barrier material selection
These solutions support aseptic processing, cold-chain logistics, and sterile medical workflows.
Why Choose Trizen Packaging for Sterile Packaging Solutions
ISO 9001:2015–certified manufacturing
Cleanroom-compatible sterile production processes
Custom sterile thermoforming packaging solutions
Medical-grade PETG, PET/APET, PP, and HDPE materials
Zero-defect quality control and particulate management
Sterilization, sealing, and regulatory compliance support
Trizen Packaging works with pharmaceutical manufacturers, medical device companies, diagnostic providers, and healthcare institutions to deliver sterile thermoforming packaging solutions that meet global regulatory and clinical standards.
Our solutions protect product integrity, enhance patient safety, and support high-efficiency sterile manufacturing environments.